Engineering the Envelop for Precision
Vector envelope engineering for targeted in-body delivery.
- Engineered Envelop Proteins
- Affinity reagents for precise cellular targeting
- Fusogenic proteins designed to maximize delivery efficiency
For Transient and Integrative In Vivo Targeting
An advanced form of adoptive cellular immunotherapy that reprograms a patient's own immune cells to recognize and eliminate cancer.
Chimeric Antigen Receptor T-cell (CAR-T) therapy is an advanced form of adoptive cellular immunotherapy that reprograms a patient's own immune cells to recognize and eliminate cancer. T lymphocytes are genetically engineered to express synthetic receptors called chimeric antigen receptors (CARs), enabling them to specifically identify tumor-associated antigens on the surface of cancer cells.
CAR-T therapies have shown strong success in hematological cancers such as leukemia, lymphoma, and multiple myeloma, achieving high response rates in relapsed or refractory patients. Next-generation CAR-T cells aim to overcome challenges in solid tumors, including heterogeneity, limited immune infiltration, and immunosuppressive microenvironments. New designs such as multi-target, logic-gated, and armored CAR-T cells seek to improve targeting, persistence, and therapeutic efficacy.
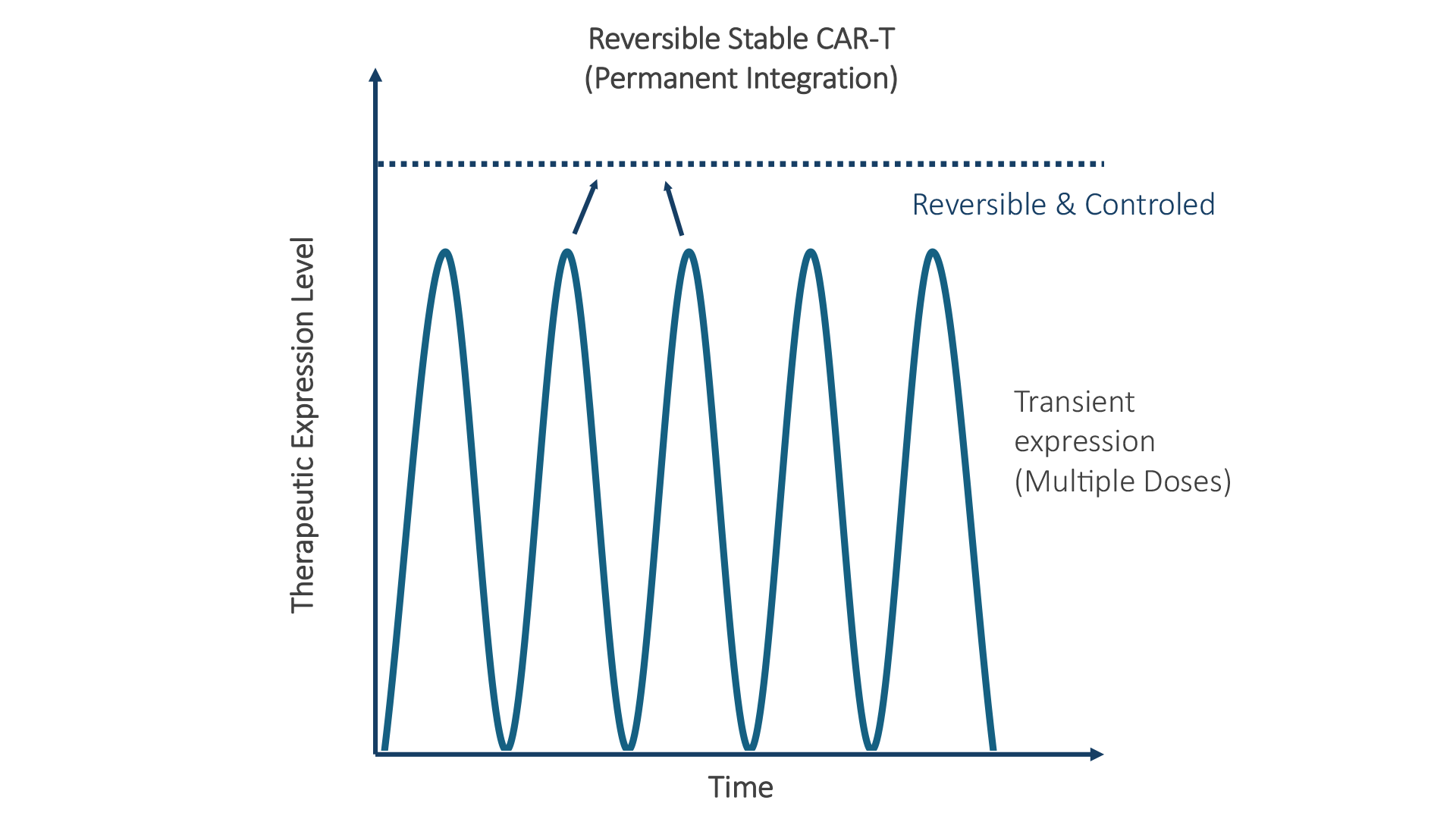
Three pillars working together: precision targeting, programmable payloads, and a non-integrative safety profile with tunable expression duration.
Vector envelope engineering for targeted in-body delivery.
A plug-and-play approach to modern medicine.
Integration versatility including:
Transforming classic medicine into modular molecular therapies.
Scalable platform applied to oncology, autoimmune diseases, and rare diseases.
Safety, control, and versatile therapeutics.
Transient and highly controllable gene expression compatible with DNA and mRNA payloads.
No genomic integration eliminates risk of mutagenesis and ensures therapeutic reversibility.
Targeted Integrative System — targetable viruses for anticancer applications and CAR-T expression and function.
Average efficacy across T-cell subpopulation switching mechanisms.
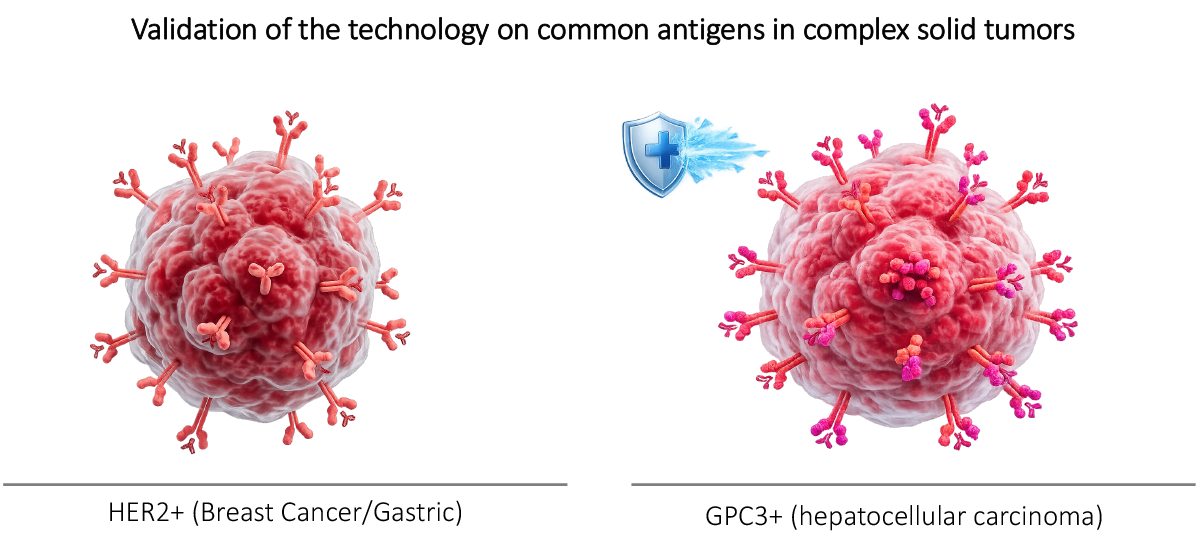
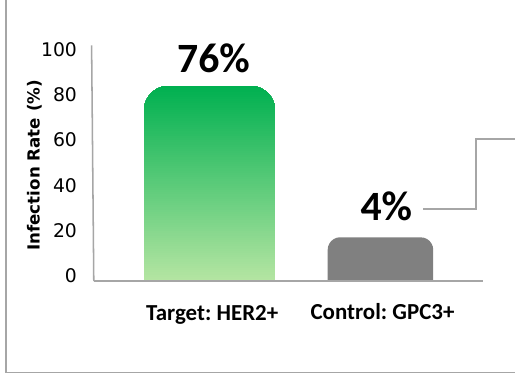
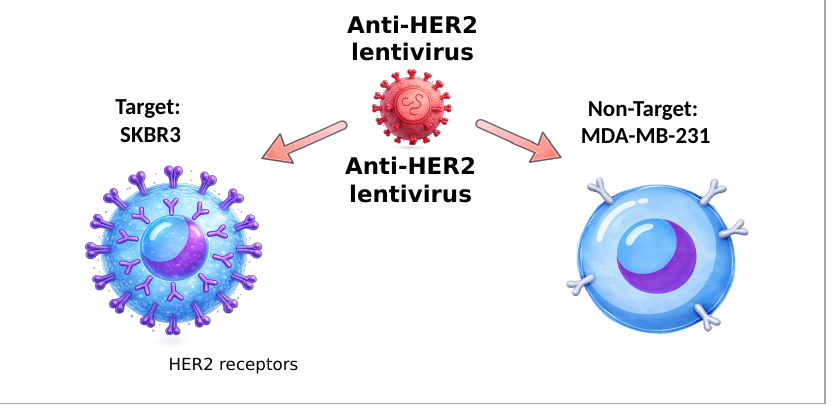
Validation on solid tumor markers HER2+ and GPC3+.

Confirmed ability to target T-cell subpopulations (CD4+ and CD8+) with distinct molecular switching mechanisms.
Validation of viral delivery on solid tumor markers (HER2+ and GPC3+).

In all four scenarios tested, unintended infection (off-target) remained consistently below 10%.
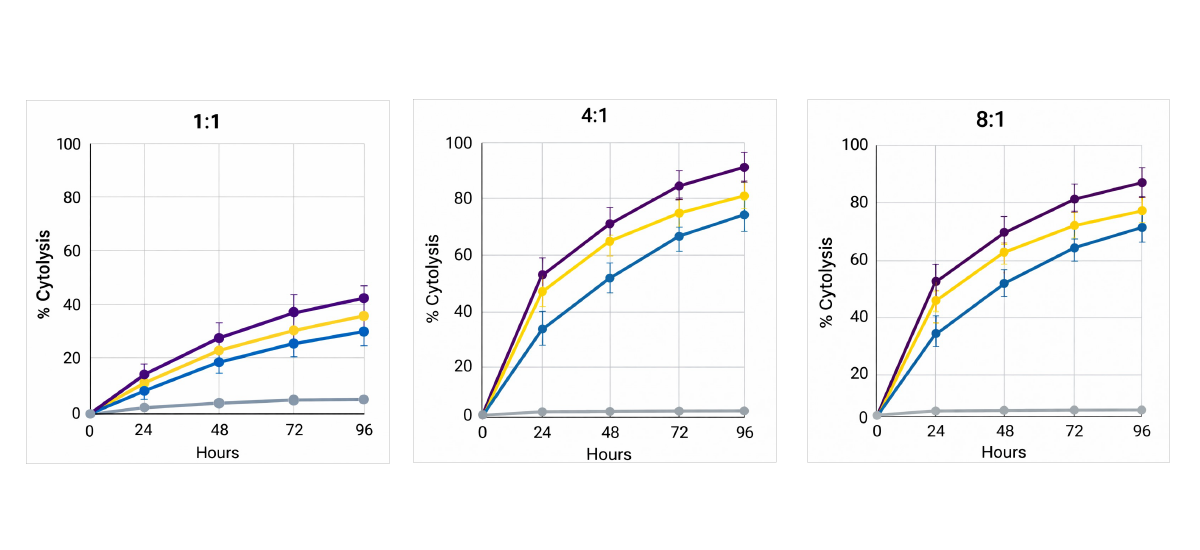
HER2+ SKBR3 cytolysis over 96 h at various target : effector ratios
Targeted Non-Integrative System — mechanisms, kinetics, and the future of tunable therapeutics.
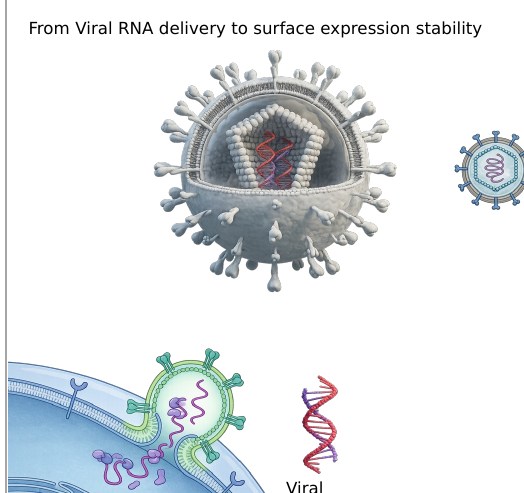
Mechanisms and kinetics of transient CAR programming, from viral RNA delivery to surface expression stability.
| Variant | Stability Profile | Day 7 Status |
|---|---|---|
| WT (Wild Type) | High Stability | Sustained (>90%) |
| TM2 | Intermediate | Moderate (~48%) |
| TM1 | Low | Minimal (~15%) |
| PM (Mutant) | Transient | Cleared (<5%) |
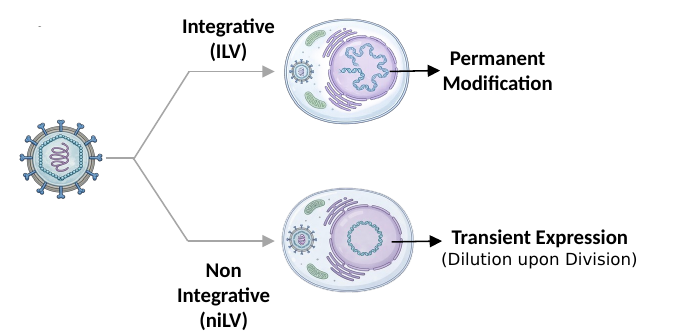
Integrative (iLV) for permanent modification vs Non-Integrative (niLV) for transient expression — choose your duration.
| Variant | Stability Profile | Day 7 Status |
|---|---|---|
| WT (Wild Type) | High Stability | Sustained (>90%) |
| TM2 | Intermediate | Moderate (~48%) |
| TM1 | Low | Minimal (~15%) |
| PM (Mutant) | Transient | Cleared (<5%) |

Transient CAR programming offers a modular platform for controlled, temporal immunotherapy.
| Variant | Stability Profile | Day 7 Status |
|---|---|---|
| WT (Wild Type) | High Stability | Sustained (>90%) |
| TM2 | Intermediate | Moderate (~48%) |
| TM1 | Low | Minimal (~15%) |
| PM (Mutant) | Transient | Cleared (<5%) |
Assessing viral integration with high-sensitivity Nested Alu-PCR — confirming the absence of mutagenic risk.

Reference. Gonçalves, J., Cardoso, M., Henrique, M., Brandão, J., & Paula, C. Exploring lentiviral platforms for transient and integrative in vivo targeting: Integrative vs non-integrative strategies for tunable CAR persistence. Poster presented at the 9th CAR-TCR Summit Europe, 2026.
Shifts CAR programming from centralized, time-intensive production toward simpler, potentially point-of-care administration.
Choice of integrative vs non-integrative delivery supports durable activity when needed, or transient, repeatable dosing for safety-sensitive use cases.
Antibody-guided tropism and cell-selective promoters can be combined to optimize who is engineered, where, and how strongly the payload is expressed.
Targeted viral delivery enables in vivo CAR-T programming with tunable persistence and an improved path to specificity and safety.